
Cellular & Molecular Biology Letters: Hsp70-Bim interaction facilitates mitophagy by recruiting parkin and TOMM20 into a complex
Authors: Ting Song, Fangkui Yin, Ziqian Wang, Hong Zhang, Peng Liu, Yafei Guo, Yao Tang & Zhichao Zhang
Abstract
Background
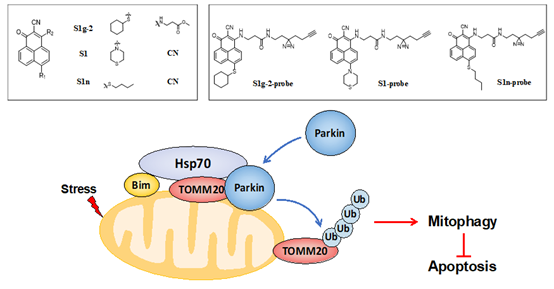
For cancer therapy, the identification of both selective autophagy targets and small molecules that specifically regulate autophagy is greatly needed. Heat shock protein 70 (Hsp70) is a recently discovered BH3 receptor that forms a protein‒protein interaction (PPI) with Bcl-2-interacting mediator of cell death (Bim). Herein, a specific inhibitor of the Hsp70-Bim PPI, S1g-2, and its analog S1, which is a Bcl-2-Bim disruptor, were used as chemical tools to explore the role of Hsp70-Bim PPI in regulating mitophagy.
Methods
Co-immunoprecipitation and immunofluorescence assays were used to determine protein interactions and colocalization patterns. Organelle purification and immunodetection of LC3-II/LC3-I on mitochondria, endoplasmic reticulum (ER) and Golgi were applied to identify specific types of autophagy. Cell-based and in vitro ubiquitination studies were used to study the role of the Hsp70-Bim PPI in parkin-mediated ubiquitination of outer mitochondrial membrane 20 (TOMM20).
Results
We found that after the establishment of their PPI, Hsp70 and Bim form a complex with parkin and TOMM20, which in turn facilitates parkin translocation to mitochondria, TOMM20 ubiquitination and mitophagic flux independent of Bax/Bak. Moreover, S1g-2 selectively inhibits stress-induced mitophagy without interfering with basal autophagy.
Conclusions
The findings highlight the dual protective function of the Hsp70-Bim PPI in regulating both mitophagy and apoptosis. S1g-2 is thus a newly discovered antitumor drug candidate that drives both mitophagy and cell death via apoptosis.
https://cmbl.biomedcentral.com/articles/10.1186/s11658-023-00458-5